FDA Reportable Food Registry
The Reportable Food Registry (RFR or the Registry) was established by Section 1005 of the Food and Drug Administration Amendments Act of 2007 (Pub. L. 110-85), which amended the Food, Drug, and Cosmetic Act (FD&C Act) by creating a new Section 417, Reportable Food Registry [21 U.S.C. 350f]. It required FDA to establish an electronic portal to which reports about instances of reportable food must be submitted to FDA within 24 hours by responsible parties and to which reports may be submitted by public health officials.
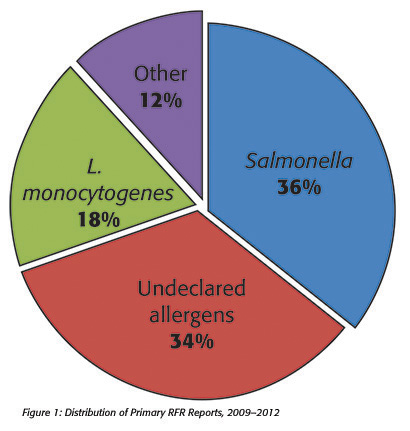
A reportable food is an article of food/feed for which there is a reasonable probability that the use of, or exposure to, such article of food will cause serious adverse health consequences or death to humans or animals.
